HydroMARK™ Plus
Breast Biopsy Site Marker
Offering the unmatched benefits of the original HydroMARK™ markers, with added features to enhance clinician confidence.
Proven HydroMARK™ Marker Performance Meets Thoughtful Innovation

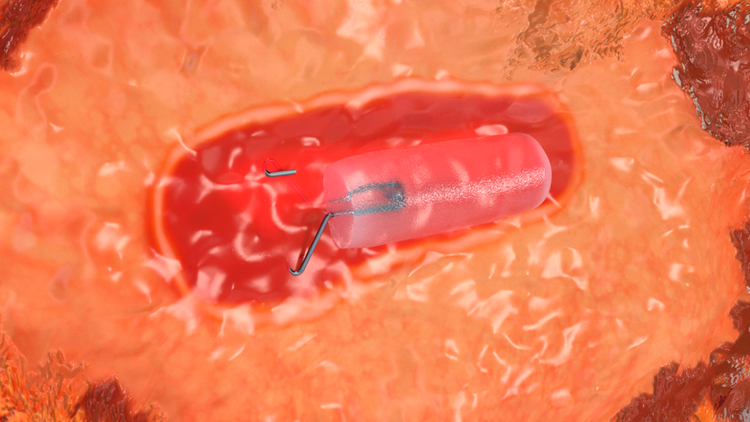
Attaches to Tissue
Exposed ‘wings’ are designed to anchor to the tissue upon deployment to mitigate displacement during surgical excision.1

Enhanced Distinguishability
The Dragonfly™ and Hummingbird™ clip shapes are distinct and discernably man-made to breast anatomy, providing enhanced distinguishability under 2D and 3D mammography.2

Visibility Upon Deployment
The titanium clips’ shape and size enhance immediate ultrasound visibility at the time of deployment.2

Same Trusted Hydrogel
Hydrogel carrier hydrates within 24 hours to provide the same unmatched ultrasound visibility as the original HydroMARK™ marker.3 Hydrogel appears strikingly anechoic under ultrasound and uniquely bright white on T2 MRI sequence.4
What Clinicians are Saying:
“Under ultrasound, the HydroMARK™ Plus marker is clearly visible as parallel hyperechoic wings, easily differentiated from other clips in the breast. This unique shape allows rapid detection in the operating room, improving efficiency and intraoperative localization.”
Priya Jadeja, MD, FACS
Breast Surgery, Oncology, Florham Park, New Jersey
Interested in Learning More?
Contact us to learn how the HydroMARK™ Plus marker can help benefit your practice.
Additional Resources
Frequently Asked Questions
Related Products

HydroMARK™ Breast Biopsy Site Marker
Enduring exclusive hydrogel technology provides long-term ultrasound visibility in percutaneous breast biopsy procedures, including axillary lymph nodes.6

LumiMARK™ Biopsy Site Marker
Three multifaceted nitinol shapes designed for easy identification form any angle.7

MammoMARK® Biopsy Site Identifier
Rapid collagen expansion anchors the marker within the biopsy cavity reducing the likelihood of movement.8-10

MammoSTAR® Biopsy Site Marker & BiomarC® Biopsy Site Marker
All-natural biopsy markers providing non-metal marking alternatives, with or without a long-lasting beta glucan carrier for unique patient sensitivities.
Would you like more information about Mammotome® Markers?
1. HydroMARK™ Plus Device Test - PCR-000299, SDR0098 Vacuum Suction Testing
2. HydroMARK™ Plus Device Test - PCR-000414, Summative Usability
3. Sakamoto, N., Fukuma, E., Tsunoda, Y. et al. Evaluation of the dislocation and long-term sonographic detectability of a hydrogel based breast biopsy site marker. Breast Cancer 25, 575–582 (2018). https://doi.org/10.1007/s12282-018-0854-8
4. Shah AD, Mehta AK, Talati N, Brem R, Margolies LR. Reprint of: Breast tissue markers: Why? What's out there? How do I choose? Clin Imaging. 2019 May-Jun;55:196-212. doi: 10.1016/j.clinimag.2019.05.014. Epub 2019 Jun 14. PMID: 31204189.
5. Indication for lymph node using HydroMARK™ Plus markers is limited to the United States and Canada, with other country registrations pending.
6. Indication may not be approved or available in your region. Please check with your local Mammotome representative.
7. LumiMARK™ Device Tests: PCR-000340 System Design, PCR-000579 Summative Usability, ES-002647 Claims Assessment
8. MammoMARK® Device Test – PRC043442 Rev B, pgs. 2, 5-6: Collagen Expansion
9. Corsi, F., Sorrentino, L., Sartani, A., Bossi, D., Amadori, R., Nebuloni, M., Truffi, M., Bonzini, M., & Foschi, D. (2015). Localization of nonpalpable breast lesions with sonographically visible clip: Optimizing tailored resection and clear margins.The American Journal of Surgery, 209(6), 950-958
10. Rosen EL, Baker JA, Soo MS. Accuracy of a collagen-plug biopsy site marking device deployed after stereotactic core needle breast biopsy. AJR Am J Roentgenol. 2003 Nov;181(5):1295-9. doi: 10.2214/ajr.181.5.1811295. PMID: 14573422.
HydroMARK™ Plus marker ultrasound photos courtesy of Dr. Robin Shermis, ProMedica Breast Care of Toledo
HydroMARK™ Plus mammography photo courtesy of Dr. Kirk Langheinz, Ochsner Lafayette General Breast Center
The Mammotome Prima™ MR system is available in the United States and Europe.
The HydroMARK™ Plus markers for MR are available in the United States.