Mammotome Launches the LumiMARK™ Biopsy Site Marker, a Tissue Marker Distinct from Every Angle
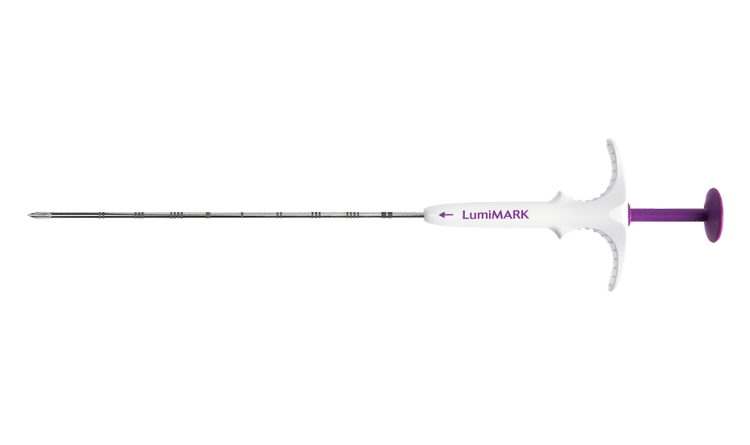
CINCINNATI, OHIO – March 28, 2024 – Mammotome announced the 510(k) clearance for the novel LumiMARK™ Biopsy Site Marker, a tissue marker line designed with visibility top of mind. The LumiMARK™ markers are distinct from every angle under ultrasound, mammography, and magnetic resonance, marking another great investment to the Mammotome® Markers portfolio.1
“Mammotome created this line of markers by collaborating with radiologists to understand their needs and concerns with current nitinol markers,” said Ben Sandefur, Mammotome Vice President and General Manager. “The LumiMARK™ Biopsy Site Markers are easily identifiable, visible under all modalities, and offer multiple contrasting shapes to support the various clinical scenarios that radiologists encounter when serving the individual needs of their patients.”
The LumiMARK™ markers are 510(k) cleared for both breast tissue and axillary lymph nodes and come in three distinctive, multifaceted 3D shapes and sizes. This provides radiologists with the power of choice and a marker to support different clinical scenarios. The unique and discernibly man-made shapes enable easy identification from any angle and quick confirmation of target tissue.1,2,3 For example, the smaller 5mm Tulip™ shape may be selected for use cases needing a smaller marker.3 The 10mm Rose™ shape is designed for effortless locating under ultrasound.3
These self-expanding markers deliver great visibility under ultrasound, appearing hyperechoic both immediately and permanently.1 The LumiMARK™ marker portfolio offers multiple three-dimensional shapes for visibility and distinguishability under ultrasound, mammography, and magnetic resonance imaging.
Mammotome continues to focus on delivering cutting-edge solutions to serve the unmet needs of clinicians and patients across the globe. The LumiMARK™ Biopsy Site Marker will be available to clinicians in the United States in the summer of 2024 with plans to expand to Canada and other select countries across the globe after regulatory approval. Click here to learn more about the LumiMARK™ marker.
Media Contact: Erica Bednar, Director of Marketing Communications
Phone: 513-391-4737
[email protected]
About Mammotome
At Mammotome, our expertise and compassion for breast care makes us the indispensable partner to physicians, clinicians and patients. Our drive for innovation is rivaled only by our compassion for the people we serve, from the clinicians and surgeons who demand consistently precise solutions, to the patients and families seeking peace of mind. We boast a comprehensive range of products that create better outcomes in breast care and provide physicians and patients with educational resources that guide their journey. Headquartered in Cincinnati, OH, Mammotome has been part of the Danaher Corporation since 2014. The Mammotome brand of products is sold in over 45 different countries throughout the world.
###
- LumiMARK™ Device Tests: PCR-000340 System Design, PCR-000579 Summative Usability, ES-002647 Claims Assessment
- LumiMARK™ Device Test: PCR-000340 System Design
- LumiMARK™ Device Tests: PCR-000579 Summative Usability and ES-002647 Claims Assessment
- LumiMARK™ Design Plan: ADD-000018, Page 6
© 2024 Devicor Medical Products, Inc. All rights reserved. Devicor and Mammotome are registered trademarks of Devicor Medical Products, Inc., in the USA and optionally in other countries. LumiMARK is a trademark of Devicor Medical Products, Inc. in the USA and optionally in other countries. Other logos, product and/or company names might be trademarks of their respective owners. MDM# 240301 Rev 3/24